
Paths to Practice Perfection
A Multi-modality Approach to Improve Healing in
a Pressure Ulcer Wound
A Multi-modality Approach to Improve Healing in
a Pressure Ulcer Wound

This is a single patient example of how using a multi-modal strategy for overcoming healing barriers in chronic wounds can be used. Single therapies frequently fail to address all the pathology found in the wound microenvironment. Recent evidence identified the importance of pathogenic biofilm in impeding wound healing: 100% of Diabetic Foot Ulcers (DFU) and over 90% of other chronic wounds contain biofilms. Agents that reverse the biofilm induced metabolic wound changes can promote healing. 1-3
As clinicians search for the key to effective biofilm destruction and the most effective combinations of therapies; questions arise about what products work best together. It is not a surprising question: clinical studies in wound care have long focused on aspects of biofilm growth as single targets of biofilm action or therapies that don’t address the triad of biofilm persistence. And, most studies fail the practical challenges faced in daily practice.
A mainstay in the treatment of pressure ulcers (PrUs), negative pressure wound therapy (NPWT), consists of placing a sponge in the wound and applying suction under an occlusive drape. NPWT facilitates wound healing in three ways. On a microscopic level the negative pressure applied to the ulcer bed induces micro-deformation of the tissue stimulating the development of granulation tissue and accelerating wound closure. On a macroscopic level the suction pulls the edges of the wound together stimulating wound contraction that promotes wound closure. Finally, the suction also removes harmful proteases that inhibit wound healing.
In some cases, when NPWT is used in PrUs the proliferation of bacteria underneath the occlusive dressing occurs. A recent patient example demonstrates how using fluorescence bacterial imaging (MolecuLight) demonstrated that large amounts of bacteria accumulated under the dressing. Bacteria are known to impede wound healing often leading to circumstances causing termination of an otherwise proven effective therapy.
This case examines the early results of a multi-patient prospective pilot study evaluating the combination of an antibiofilm agent, BlastXTM, and negative pressure wound therapy (NPWT; VAC®) after 4-weeks of active therapy. The study enrolled patients with complex long-standing pressure ulcers, a population not included in traditional clinical trials, who had failed standard of care and previous NPWT. The investigators hypothesized that adding an antibiofilm agent to the wound bed under NPWT would decrease bacterial burden, reduce proteases, and promote wound healing.
Patients returned to the clinic once per week for sharp debridement, measurement of bacterial bio load, bacterial fluorescence and wound area measurement. Additional dressing changes were performed by trained staff to maintain study guidelines.

This confirming result presented here suggests that adding BlastX to NPWT may improve wound healing trajectories and facilitate healing, but indeed reduced the bacteria bioburden under the NPWT dressing in the wound and reduce the number of proteases indicating that the negative impact of biofilm was alleviated.
Biofilm is built like a fortress and unless the building blocks of biofilm (extracellular polymeric substance/matrix [EPS]) is deconstructed, the structure continues to protect and nurture the bacteria within the EPS even though the free-floating bacteria may be destroyed. With so many products in our wound healing toolbox, and amid different biofilm narratives, it is often difficult for clinicians to choose optimal multi-modal therapies. The combination of NPWT and BlastX appears to be an effective and efficient treatment option that does not lead to adverse events. It has become more evident that treating biofilm should be an early consistent standard treatment for all wounds.
70-year-old male with Multiple Sclerosis and limited mobility. Full thickness Pressure Ulcer of 17 months duration. Previous failed 2-month course of Negative Pressure Wound Therapy about six months prior to beginning of this trial. Positive bacterial protease (BPA) and elevated protease activity (EPA) markers.
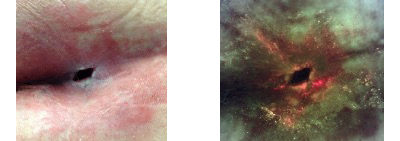
End of study: 50% reduction in wound surface area. iX Image is now negative even for the peri-wound skin. Proteases negative.
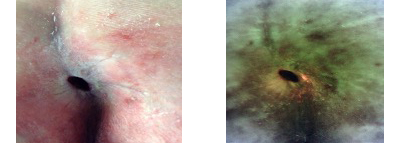
-
Attinger C, Wolcott R. Clinically Addressing Biofilm in Chronic Wounds. Adv Wound Care. 2012;1(3):127-132.
-
Johani K, Malone M, Jensen S, Gosbell I, Dickson H, Hu H, Vickery K. Microscopy visualisation confirms multi-species biofilms are ubiquitous in diabetic foot ulcers. Int Wound J. 2017; doi: 10.1111/iwj.12777
-
Snyder RJ, Bohn G, Hanft J, Harkless L, Kim P, Lavery L, Schultz G, Wolcott R. Wound Biofilm: Current Perspectives and Strategies on Biofilm Disruption and Treatments. Wounds: a Compendium of Clinical Research and Practice [01 Jun 2017, 29(6): S1-S17]


Comments
There are 0 comments for this article